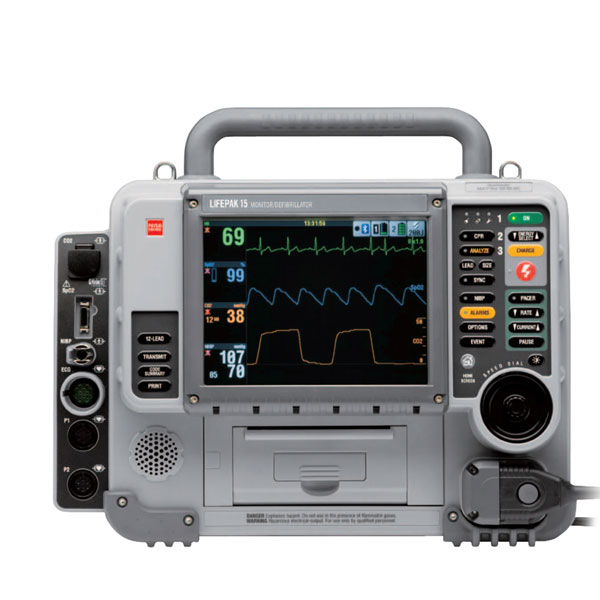
Physio Control Lifepak 15
Certified Refurbished Medical Equipment!
The mentioned price is for a Lifepak 15 basic option. We have the loaded unit in stock as well. Please call us for a discounted price.
The Physio-Control Lifepak 15 defibrillator/monitor gives you more monitoring capabilities than any other defibrillator/monitor. The Lifepak 15 continuously monitors all 12-leads (OPTIONAL) in the background and alerts the operator to any changes, gives EtCO2 (OPTIONAL) with continuous waveform capture, and features Masimo Rainbow technology to help detect hard-to-diagnose conditions with noninvasive monitoring of carbon monoxide, SpO2, and methemoglobin. Using the CODE-STAT on the Lifepak 15 data review software, the Lifepak Defibrillator Model 15 allows the operator to transmit, trend, or display the recorded data for post-event review.
$20,615.00
Features
The Physio-Control Lifepak 15 defibrillator/monitor gives you more monitoring capabilities than any other defibrillator/monitor. The Lifepak 15 continuously monitors all 12-leads in the background and alerts the operator to any changes, gives EtCO2 with continuous waveform capture, and features Masimo Rainbow technology to help detect hard-to-diagnose conditions with noninvasive monitoring of carbon monoxide, SpO2, and methemoglobin. Using the CODE-STAT on the Lifepak 15 data review software, the Lifepak Defibrillator Model 15 allows the operator to transmit, trend, or display the recorded data for post-event review.
ADAPTIV Biphasic Technology Manual; AED External Defibrillation
8.4” Color LCD Screen
CPR Metronome uses audible prompts to guide you without distracting vocal critique
Noninvasive Pacing (Demand & Non-Demand)
12-Lead ECG capability
100mm Printer
A lithium-ion dual battery system
Handle that fits two gloved hands for easy pass off.
Advanced Monitoring Parameters
With more monitoring capabilities than any other monitor/defibrillator, the 15 gives you EtCO2 with continuous waveform capture. Masimo Rainbow technology helps you detect hard-to-diagnose conditions and improve patient care with noninvasive monitoring of carbon monoxide, SpO2, and methemoglobin. In addition, the 15 offers temperature monitoring—and like other data, you can transmit it to other systems, trend it, or display for post-event review in CODE-STAT data review software.
Advanced Support for Treating Cardiac Patients
The 15 continuously monitors all 12 leads in the background and alerts you to changes using the ST-Segment trend monitoring feature, after acquiring the initial 12-lead. Additionally, STJ values are included on the 12-lead printout to help you identify changes. The 15 also works seamlessly with the web-based LIFENET System 5.0, so you can automatically share critical patient data with multiple patient care teams.
Full Energy Up To 360 Joules, for Every Patient Who Needs It
The Lifepak 15 monitor/defibrillator features 360J biphasic technology, which gives you the option of escalating your energy dose up to 360J for difficult-to-defibrillate patients. Why is this necessary? Recent studies have shown that defibrillation is common among VF cardiac arrest patients and that defibrillation of recurring episodes of VF is increasingly difficult. A randomized controlled clinical trial shows the rate of VF termination was higher with an escalating higher energy regimen of 200J and over.
Proven CPR Guidance and Post Event Review
The CPR Metronome in the Lifepak 15 monitor uses audible prompts to guide you without distracting vocal critique. A metronome has been a feature that has been demonstrated to help professionals perform compressions and ventilation within the recommended range of the 2015 AHA Guidelines. Post-event review of CPR data and delivering feedback to the team has been shown to be effective in improving CPR quality in both hospital and out-of-hospital.2,3,4 And by transmitting code data directly to CODE-STAT Data Review software, EMS personnel can review CPR statistics and provide training and feedback where it is most needed.
Specifications
General
This Lifepak defibrillator has six main operating modes:
AED Mode: for automated ECG analysis and a prompted treatment protocol for patients in cardiac arrest.
Manual Mode: for performing manual defibrillation, synchronized cardioversion, noninvasive pacing, and ECG and vital sign monitoring.
Archive Mode: for accessing stored patient information.
Setup Mode: for changing default settings of the operating functions.
Service Mode: for authorized personnel to perform diagnostic tests and calibrations.
Demo Mode: for simulated waveforms and trend graphs for demonstration purposes.
Physical Characteristics
Weight: Basic monitor/defibrillator with new roll paper and two batteries installed: 7.9 kg (17.5 lb) Fully featured monitor/defibrillator = with new roll paper and two batteries installed: 8.4 kg (18.5 lb)
Lithium-ion battery: ≤0.6kg (1.3lb)
Accessory Bags and Shoulder Strap: 1.77 kg (3.9 lb)
Standard (hard) Paddles: 0.95 kg (2.1 lb)
Height: 31.7 cm (12.5 in)
Width: 40.1 cm (15.8 in)
Depth: 23.1 cm (9.1 in)
Display
Size (active viewing area): 212 mm (8.4 in) diagonal; 171 mm (6.7 in) wide x 128 mm (5.0 in) high
Resolution: display type 640 dot x 480 dots color backlit LCD
User Selectable Display Mode: full color or SunVue™ display high contrast
Display: a minimum of 5 seconds of ECG and alphanumerics for values, device instructions, or prompts
Display: up to three waveforms
Waveform Display Sweep Speed: 25 mm/sec for ECG, Sp02, IP, and 12.5 mm/sec for CO2
Data Management
The device captures and stores patient data, events (including waveforms and annotations), and continuous waveform and patient impedance records in internal memory. The user can select and print reports, and transfer the stored information via supported communication methods
Report Types:
Three format types of CODE SUMMARY™ critical event record: short, medium, and long
12-lead ECG with STEMI statements
Continuous Waveform (transfer only)
Trend Summary
Vital Sign Summary
Snapshot
Memory Capacity:
Total capacity is 360 minutes of continuous ECG, 90 minutes of continuous data from all channels, or 400 single waveform events.
Maximum memory capacity for a single patient includes up to 200 single waveform reports and 90 minutes of continuous ECG
Communications
The device is capable of transferring data records by wired or wireless connection. This device complies with Part 15 of the FCC rules, and its operation is subject to the following two conditions: (1) this device may not cause harmful interference, and (2) this device must accept any interference received, including interference that may cause undesired operation.
Serial Port RS232 communication + 12V available
Limited to devices drawing a maximum of 0.5 A current
Bluetooth® technology provides short-range wireless communication with other Bluetooth-enabled devices
Monitor
ECG
ECG is monitored via several cable arrangements:
3-wire cable is used for 3-lead ECG monitoring.
A 5-wire cable is used for 7-lead ECG monitoring.
A 10-wire cable is used for 12-lead ECG acquisition. When the chest electrodes are removed, the 10-wire cable functions as a 4-wire cable.
Standard paddles or QUIK-COMBO pacing/defibrillation/ECG electrodes are used for paddles lead monitoring.
Frequency Response:
Monitor: 0.5 to 40 Hz or 1 to 30 Hz
Paddles: 2.5 to 30 Hz
12-lead ECG diagnostic: 0.05 to 150 Hz
Lead Selection:
Leads I, II, III, (3-wire ECG cable)
Leads I, II, III, AVR, AVL, and AFV acquired simultaneously (4-wire ECG cable)
Leads I, II, III, AVR, AVL, AFV, and C lead acquired simultaneously (5-wire ECG cable)
Leads I, II, III, AVR, AVL, AFV, V1,V2,V3,V4,V5, and V6 acquired simultaneously (10-wire ECG cable)
ECG size:
4, 3, 2.5, 2, 1.5, 1, 0.5, 0.25 cm/mV (fixed at 1 cm/mV for 12-lead)
Heart Rate Display:
20–300 bpm digital display
Accuracy: ±4% or ±3 bpm, whichever is greater
QRS Detection Range Duration: 40 to 120 msec
Amplitude: 0.5 to 5.0 m
Common Mode Rejection (CMRR):
ECG Leads: 90 dB at 50/60 Hz